The MASTERGRAFT
® Family of Products is biphasic, resorbable ceramics composed of hydroxyapatite (HA) and beta-tricalcium phosphate (β-TCP), either in granule form or distributed evenly throughout a Type I collagen in putty form. The biphasic blend of HA/β-TCP throughout the products provides a purely osteoconductive graft made of materials similar to the mineral found inherently in bone.
1,2 The uniform mixture of hydroxyapatite and beta-tricalcium phosphate provides the surgeon with characteristics that balance long-term stability with a natural cell-mediated (osteoclastic) resorption process.
2
MASTERGRAFT® Granules and MASTERGRAFT® Mini Granules
Composition, Structure, and Handling
MASTERGRAFT
® Granules and MASTERGRAFT
® Mini Granules are biphasic, resorbable, ceramic granules composed of 15% hydroxyapatite (HA) and 85% betatricalcium phosphate (β-TCP). The granules have a natural, interconnected, porous structure which mimics the natural structure of bone and allows for rapid, homogenous bone ingrowth throughout each granule.
2 Each granule is 80% porous with an average pore size of 500 microns and 125 microns interconnected diameter. MASTERGRAFT
® Granules have an average diameter of 1.6mm – 3.2mm. MASTERGRAFT
® Mini Granules have an average diameter of 0.5mm – 1.6mm.
Through a highly porous granular structure and the 15% HA/85% β-TCP chemical composition, MASTERGRAFT
® Granules and MASTERGRAFT
® Mini Granules facilitate rapid, homogenous osseointegration, which supports the bone healing process by acting as a scaffold over which bone can grow. The porosity of the material provides an excellent basis for vascularization and penetration of associated cells, which support integration of the substitute materials required for healing while preserving the bony architecture and attached gingiva.
4
Average Pore Size:
MASTERGRAFT® Granule
25 X magnification
125 microns
500 microns
Summary of Indications
MASTERGRAFT® Granules can be used alone or in combination with autogenous bone in oral/maxillofacial augmentation or reconstruction and other larger bony defects. MASTERGRAFT® Mini Granules are ideal for smaller defects such as extraction site preservation and periodontal defects, or in combination with autogenous bone in larger defects such as sinus augmentation and cystic defects.
MASTERGRAFT® Putty
Composition, Structure, and Handling
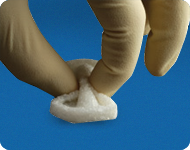
MASTERGRAFT
® Putty is composed of resorbable ceramic granules uniformly distributed through a highly purified resorbable Type I bovine collagen. The resorbable ceramic granules of 15% HA, 85% β-TCP balance long-term stability with complete resorption.
2 The granules range in size from 0.5mm-1.6mm in diameter and are 80% porous. The Type I bovine collagen used is composed of two formulations of collagen: 70% insoluble fibrous collagen and 30% soluble collagen. The collagen allows the graft materials to be malleable and resistant to water solubility while still maintaining the graft's integrity. MASTERGRAFT
® Putty is an osteoconductive scaffold that resorbs by cell-mediated creeping substitution, and is gradually replaced with new, homogenous bone growth throughout the site. MASTERGRAFT
® Putty also enables the user to perform radiographic assessment of the graft after implantation.
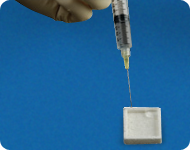
MASTERGRAFT
® Putty was designed for easy hydration using sterile water or blood. Once hydrated, it can be mixed with autogenous bone to increase the volume of graft material while providing a small level of osteogenic potential due to the presence of autograft.
3




.jpg)


.jpg)



.png)








.jpg)



 İmplant
İmplant
 Ortodonti
Ortodonti
 Tranier
Tranier
 Disposable
Disposable